Abstract
Goals of work
Patients with low-risk neutropenic fever as defined by the Multinational Association of Supportive Care in Cancer (MASCC) score might benefit from ambulatory treatment. Optimal management remains to be clearly defined, and new oral antibiotics need to be evaluated in this setting.
Materials and methods
Cancer patients with febrile neutropenia and a favorable MASCC score were randomized between oral moxifloxacin and intravenous ceftriaxone. All were fit for early hospital discharge. The global success rate was related to the efficacy of monotherapy, as well as to the success of ambulatory monitoring.
Main results
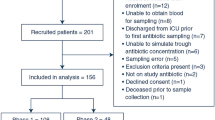
The trial was closed prematurely because of low accrual. Ninety-six patients were included (47 in the ceftriaxone arm and 49 in the moxifloxacin arm). A total of 65% were women, 30.2% had lymphoma, 34.4% had metastatic, and 35.4% had non-metastatic solid tumors. The success rates of home antibiotics were 73.9% and 79.2% for ceftriaxone and moxifloxacin, respectively. Seven patients were not discharged, and 14 required re-hospitalization. There were 17% of microbiologically documented infections that were, in most cases, susceptible to oral monotherapy.
Conclusions
These results suggest that MASCC is a valid and useful tool to select patients for ambulatory treatments and that oral moxifloxacin monotherapy is safe and effective for the outpatient treatment of cancer patients with low-risk neutropenic fever.
Similar content being viewed by others
References
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner H, Fleishman SB, de Haes JC et al (1993) The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 85:365–376
Aktas Z, Gonullu N, Salcioglu M, Bal C, Ang O (2002) Moxifloxacin activity against clinical isolates compared with the activity of ciprofloxacin. Int J Antimicrob Agents 20:196–200
Balfour JA, Wiseman LR (1999) Moxifloxacin. Drugs 57:363–373
Biron P, Fuhrmann C, Cure H, Viens P, Lefebvre D, Thyss A, Viot M, Soler-Michel P, Rollin C, Gres JJ (1998) Cefepime versus imipenem-cilastatin as empirical monotherapy in 400 febrile patients with short duration neutropenia. CEMIC (Study Group of Infectious Diseases in Cancer). J Antimicrob Chemother 42:511–518
Chamilos G, Bamias A, Efstathiou E, Zorzou PM, Kastritis E, Kostis E, Papadimitriou C, Dimopoulos MA (2005) Outpatient treatment of low-risk neutropenic fever in cancer patients using oral moxifloxacin. Cancer 103:2629–2635
Chamorey E, Magne N, Foa C, Otto J, Largillier R, Viot M, Benard-Thiery I, Thyss A (2004) Ceftriaxone monotherapy for the treatment of febrile neutropenia in patients with solid tumors: a prospective study of 100 episodes. Med Sci Monit 10:I119–I125
Escalante CP, Rubenstein EB, Rolston KV (1997) Outpatient antibiotic therapy for febrile episodes in low-risk neutropenic patients with cancer. Cancer Invest 15:237–242
Escalante CP, Weiser MA, Manzullo E, Benjamin R, Rivera E, Lam T, Ho V, Valdres R, Lee EL, Badrina N, Fernandez S, DeJesus Y, Rolston K (2004) Outcomes of treatment pathways in outpatient treatment of low risk febrile neutropenic cancer patients. Support Care Cancer 12:657–662
Freifeld A, Marchigiani D, Walsh T, Chanock S, Lewis L, Hiemenz J, Hiemenz S, Hicks JE, Gill V, Steinberg SM, Pizzo PA (1999) A double-blind comparison of empirical oral and intravenous antibiotic therapy for low-risk febrile patients with neutropenia during cancer chemotherapy. N Engl J Med 341:305–311
Hidalgo M, Hornedo J, Lumbreras C, Trigo JM, Colomer R, Perea S, Gomez C, Ruiz A, Garcia-Carbonero R, Cortes-Funes H (1999) Outpatient therapy with oral ofloxacin for patients with low risk neutropenia and fever: a prospective, randomized clinical trial. Cancer 85:213–219
Hughes WT, Armstrong D, Bodey GP, Bow EJ, Brown AE, Calandra T, Feld R, Pizzo PA, Rolston KV, Shenep JL, Young LS (2002) 2002 guidelines for the use of antimicrobial agents in neutropenic patients with cancer. Clin Infect Dis 34:730–751
Innes HE, Smith DB, O'Reilly SM, Clark PI, Kelly V, Marshall E (2003) Oral antibiotics with early hospital discharge compared with in-patient intravenous antibiotics for low-risk febrile neutropenia in patients with cancer: a prospective randomised controlled single centre study. Br J Cancer 89:43–49
Jun HX, Zhixiang S, Chun W, Reksodiputro AH, Ranuhardy D, Tamura K, Matsumoto T, Lee DG, Purushotaman SV, Lim V, Ahmed A, Hussain Y, Chua M, Ong A, Liu CY, Hsueh PR, Lin SF, Liu YC, Suwangool P, Jootar S, Picazo JJ (2005) Clinical guidelines for the management of cancer patients with neutropenia and unexplained fever. Int J Antimicrob Agents 26:S128–S132
Karthaus M, Egerer G, Kullmann KH, Ritter J, Jurgens H (1998) Ceftriaxone in the outpatient treatment of cancer patients with fever and neutropenia. Eur J Clin Microbiol Infect Dis 17:501–504
Kern WV, Cometta A, De Bock R, Langenaeken J, Paesmans M, Gaya H (1999) Oral versus intravenous empirical antimicrobial therapy for fever in patients with granulocytopenia who are receiving cancer chemotherapy. International Antimicrobial Therapy Cooperative Group of the European Organization for Research and Treatment of Cancer. N Engl J Med 341:312–318
Klaassen RJ, Goodman TR, Pham B, Doyle JJ (2000) “Low-risk” prediction rule for pediatric oncology patients presenting with fever and neutropenia. J Clin Oncol 18:1012–1019
Klastersky J, Paesmans M, Rubenstein EB, Boyer M, Elting L, Feld R, Gallagher J, Herrstedt J, Rapoport B, Rolston K, Talcott J (2000) The Multinational Association for Supportive Care in Cancer risk index: a multinational scoring system for identifying low-risk febrile neutropenic cancer patients. J Clin Oncol 18:3038–3051
Klastersky J, Paesmans M, Georgala A, Muanza F, Plehiers B, Dubreucq L, Lalami Y, Aoun M, Barette M (2006) Outpatient oral antibiotics for febrile neutropenic cancer patients using a score predictive for complications. J Clin Oncol 24:4129–4134
Malik IA, Khan WA, Karim M, Aziz Z, Khan MA (1995) Feasibility of outpatient management of fever in cancer patients with low-risk neutropenia: results of a prospective randomized trial. Am J Med 98:224–231
Mullen CA, Petropoulos D, Roberts WM, Rytting M, Zipf T, Chan KW, Culbert SJ, Danielson M, Jeha SS, Kuttesch JF, Rolston KV (1999) Outpatient treatment of fever and neutropenia for low risk pediatric cancer patients. Cancer 86:126–134
Petrilli AS, Dantas LS, Campos MC, Tanaka C, Ginani VC, Seber A (2000) Oral ciprofloxacin vs. intravenous ceftriaxone administered in an outpatient setting for fever and neutropenia in low-risk pediatric oncology patients: randomized prospective trial. Med Pediatr Oncol 34:87–91
Rolston KV (2004) The Infectious Diseases Society of America 2002 guidelines for the use of antimicrobial agents in patients with cancer and neutropenia: salient features and comments. Clin Infect Dis 39:S44–S48
Rolston K, Rubenstein E, Elting L, Escalante E, Manzullo E, Bodey GP (1995) Ambulatory management of febrile episodes in low-risk neutropenic patients. In: Programs and abstracts of the 35th interscience conference on antimicrobial agents and chemotherapy, San Francisco. Abstract LM81. American Society for Microbiology, Washington, DC, USA
Rubenstein EB, Rolston KV (1995) Outpatient treatment of febrile neutropenic patients with cancer. Eur J Cancer 31A:2–4
Samonis G, Anaissie E, Kalbakis K, Georgoulias V (1997) Therapy for low risk cancer patients with fever and neutropenia:results of a prospective randomized trial with cost analysis. Programs and abstracts of the 37th interscience conference on antimicrobial agents and chemotherapy, Toronto, Canada. Abstract LM51. American Society for Microbiology, Washington, DC, USA
Sebban C, Fuhrmann C, Perol D, Devaux Y, Ghesquiere H, Galand-Desme S, Leveque N, Claude L, Biron P (2006) Prise en charge à domicile des neutropénies fébriles courtes après chimiothérapie: résultats et leçons d’une étude pilote menée sur deux ans dans un centre de lutte contre le cancer. Oncologie 8:HS29–HS33
Talcott JA, Siegel RD, Finberg R, Goldman L (1992) Risk assessment in cancer patients with fever and neutropenia: a prospective, two-center validation of a prediction rule. J Clin Oncol 10:316–322
Uys A, Rapoport BL, Anderson R (2004) Febrile neutropenia: a prospective study to validate the Multinational Association of Supportive Care of Cancer (MASCC) risk-index score. Support Care Cancer 12:555–560
Vidal L, Paul M, Ben Dor I, Soares-Weiser K, Leibovici L (2004) Oral versus intravenous antibiotic treatment for febrile neutropenia in cancer patients: a systematic review and meta-analysis of randomized trials. J Antimicrob Chemother 54:29–37
Viscoli C, Bruzzi P, Castagnola E, Boni L, Calandra T, Gaya H, Meunier F, Feld R, Zinner S, Klastersky J, Glauser M (1994) Factors associated with bacteraemia in febrile, granulocytopenic cancer patients. The International Antimicrobial Therapy Cooperative Group (IATCG) of the European Organization for Research and Treatment of Cancer (EORTC). Eur J Cancer 30:430–437
Acknowledgement
This study was supported by a grant from Bayer Pharma France. We thank Dr. Pierre Arvise for the logistical and technical assistance; Giovanna Gonnon, Séverine Guillemaut, and Mathieu Cassin for the help in the study design, study monitoring, and data analysis; Carlos Poncelas for the help in the management of outpatient treatment; and Marie-Dominique Reynaud for the editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sebban, C., Dussart, S., Fuhrmann, C. et al. Oral moxifloxacin or intravenous ceftriaxone for the treatment of low-risk neutropenic fever in cancer patients suitable for early hospital discharge. Support Care Cancer 16, 1017–1023 (2008). https://doi.org/10.1007/s00520-007-0383-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-007-0383-z